Atomic radius of bromine12/18/2023 Also replace URL for the actual url of this page (The stay, ok?). Now replace dd, mmmm and yyyy with the day, month, and year you browsed this page. "Ionic Radius of Bromine (Br) [& Discovery, Color, Uses. To make your life (and citation) easier just copy & paste the information below into your assignment or essay: That gives credibility to your paper and it is sometimes required in higher education. When you need to include a fact or piece of information in an assignment or essay you should also include where and how you found that piece of information. How about an incentive to share this post? (You will help other colleagues find this blog)ĭownload and enjoy this complete and colored periodic table for you to edit and enjoy. Need an editable periodic table to edit? Maybe add your school logo, work team or anything else to make your paper look cool?Īlong with basic atom / element information (like Bromine ionic radius and all the other atomic data), it also comes with color coded info about: State (Gas, Liquid or Solid at room temperature), Groups/series details and much more. How a small number of atoms can be joined and form completely different substances.Want to learn more details and data about Bromine (Br)? Check my Elements Comprehensive List.Īre you having trouble understanding the basics of atomic elements? This video will walk you through: Now it is primarily used in dyes, disinfectants, and photographic chemicals. It was once used in large quantities to make a compound that removed lead compound build up in engines burning leaded gasoline. In the case of Bromine the ionic radius is 1.96 (-1) Å. Speaking of Bromine, let me walk you through some interesting details. When an electron is added, a new proton is also added to the nucleus, which. Question: Question 12 0 / 1 point The atomic radius of bromine is smaller than the atomic radius of selenium because there are more energy levels in. Within a period of elements, each new electron is added to the same shell. The first atomic radius periodic trend is that atomic size decreases as you move left to right across a period. Ok so what is the ionic radius of a Bromine ion? Atomic Radius Trend 1: Atomic Radii Decrease From Left to Right Across a Period. Therefore, there are various non-equivalent definitions of atomic radius.All atom's ions have an ionic radius, even Bromine. group number 17 but atomic radius increases down the group in the periodic table hence chlorine is. However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. While chlorine and bromine are present in the same group i.e. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. All the elements of similar categories show a lot of similarities and differences in their chemical, atomic, physical properties and uses. Compare elements on more than 90 properties. It must be noted, atoms lack a well-defined outer boundary. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. Bromine and Chlorine on the basis of their properties, attributes and periodic table facts. 
The atomic radius of Bromine atom is 120pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. 
The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Bromine are 79 81. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. 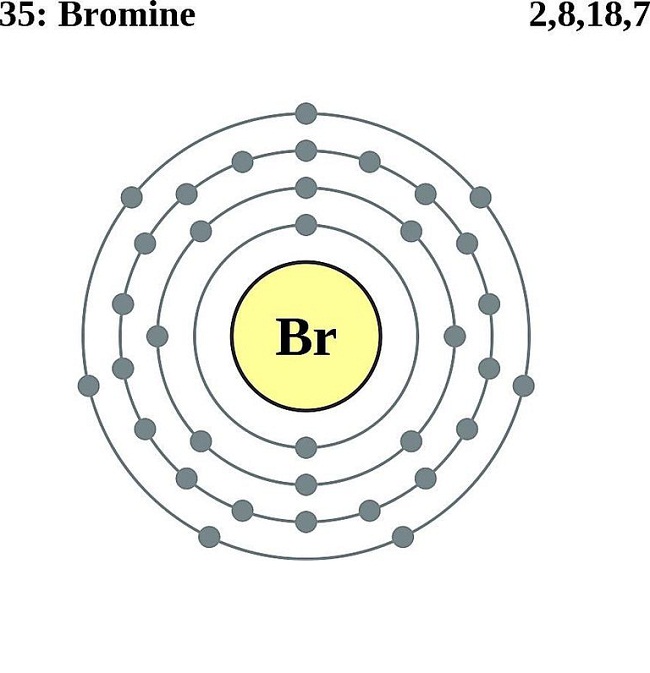
The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Atomic Number – Protons, Electrons and Neutrons in Bromineīromine is a chemical element with atomic number 35 which means there are 35 protons in its nucleus.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
